Drugs Slip Past FDA, Sell Unapproved by the MillionsBy Justin BlumBloomberg Oct. 12, 2006 |
Popular 
FL State Sen. Randy Fine Celebrated Israel Killing an American - Trump Just Endorsed Him For Congress
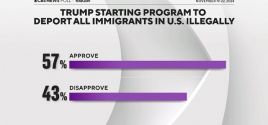
Poll: 57% of Americans Support Trump Starting Program to Deport All Illegal Immigrants

Trump Nominates Pam Bondi for Attorney General

Netanyahu Cries 'Antisemitism' After International Criminal Court Issues Warrant for His Arrest

Matt Gaetz Withdraws from Consideration as Attorney General
 Oct. 11 (Bloomberg) -- Ballay Pharmaceuticals Inc. had a choice nine years ago: seek U.S. approval to sell a prescription decongestant or slip the drug on the market and hope regulators wouldn't order it off. The company chose to market the medicine, Balamine DM, to doctors and pharmacies without the consent of the U.S. Food and Drug Administration, and the agency didn't stop Wimberley, Texas-based Ballay. Almost 2 percent of U.S. prescriptions dispensed last year, or as many as 73 million, were for unapproved medicines such as Balamine, the FDA estimates. After years of paying little attention, the FDA now says it will step up efforts to get many of the drugs approved or taken off the market. While some medications may be lawful because they are safe and effective, the FDA says the majority are illegal -- and some may be killing or injuring people. ``It's a tremendous risk, and the problem is that the public doesn't know about it,'' says Larry D. Sasich, a professor of pharmacy practice at Lake Erie College of Osteopathic Medicine in Erie, Pennsylvania. Sasich has written about the hazards of pharmaceuticals for Public Citizen, a Washington-based advocacy group. The makers of unsanctioned medicines, often closely held companies with fewer than 500 employees, defend their practices as legal. The law allows companies to market drugs that are ``generally recognized'' as effective and safe. Internet Ads To gain explicit clearance, companies must submit studies proving that products work and are safe. Some companies do register drugs with the FDA when they market them, making the agency aware of the products while skipping the approval process. Marketers promote unapproved drugs through the Internet, ads in journals and sales visits to doctors. The names and packaging of the products -- such as Cortane-B to treat ear infections --don't reveal the unapproved status. Cortane-B's maker, Blansett Pharmacal Co. in North Little Rock, Arkansas, says the product is safe and effective yet the company plans to seek FDA approval. Physicians prescribe unapproved drugs and pharmacies sell them often without knowing their status. Insurance plans, including some tied to the U.S. Medicare program for the disabled and elderly, often pay for unapproved medicines -- sometimes knowingly. It is legal to prescribe, fill prescriptions for, and insure unapproved drugs. FDA's `Renewed Emphasis' The FDA hasn't developed a list of unapproved drugs and says it doesn't know whether the number -- which it estimates to be in the thousands -- is increasing. The agency plans to place ``renewed emphasis'' on the drugs, says Deborah M. Autor, director of the office of compliance at the FDA. ``We are committed to ensuring that all drugs in the U.S. meet safety and effectiveness standards,'' she says. Representative Edward Markey, a Democrat from Massachusetts who is frequently critical of the FDA, says the agency isn't doing enough. ``The FDA should not sit idly by while companies market products that could be worse than snake oil,'' he says. 21 Deaths As of April, Balamine and more than 100 other unapproved medicines contained an ingredient associated with the deaths of 21 children under 2 years old during the past 23 years, according to the FDA. The substance is carbinoxamine, an antihistamine. While ``it is not clear that the carbinoxamine caused these deaths,'' the FDA said in a June statement, the agency ordered companies to stop making unapproved drugs with the compound. Ballay and other drugmakers say they stopped production of carbinoxamine-containing medicines or removed the ingredient after the FDA's statement. The products had been on the market for years and supplies may still be in pharmacies, as the FDA didn't demand the manufacturers recall the products. Balamine met a medical need, says Terry Ballay, president of Ballay, which is closely held and has fewer than 20 employees. ``I'm not a villain,'' he says. ``I'm really not. I think I help society on the whole.'' Paid Listing Ballay paid for a listing for Balamine and another drug in this year's ``Physicians' Desk Reference,'' an annual directory used by many doctors to research drugs. Companies listing products in the book aren't required to demonstrate that the drugs have been approved by the FDA, says Kyle Christensen, a spokesman for the publisher, a unit of Thomson Corp. According to Ballay's entry, Balamine drops can be used in children as young as 1 month and Balamine syrup in infants at least 18 months old. The appropriate dose of carbinoxamine for children under 2 years hasn't been established, the FDA says. Ballay declines to explain why he didn't apply for FDA approval to sell Balamine before he put it on the market in 1997. ``We sold a lot of it, and the doctors loved it,'' he says, refusing to divulge figures. He says he knows of no injuries caused by the drug. Mixtures Like Balamine, other unapproved drugs often contain mixtures of ingredients that were included in medications that had previously gained U.S. clearance. New doses, combinations and prescribing instructions found in the unapproved versions may pose risks, according to the FDA and independent specialists. ``You just don't know what contaminant might be lurking in one of these compounds or whether there's a new safety risk in one of these combinations,'' says Lee Vermeulen, a professor at the University of Wisconsin-Madison School of Pharmacy. Drugs lacking an FDA imprimatur have come to public attention before. In 1984, the FDA linked 38 infant deaths to an unapproved intravenous vitamin E solution used to treat premature babies. The drug was withdrawn. The FDA says it has taken ``numerous'' actions during the past two decades to spur compliance with the law. Officials haven't responded to requests for details. Warning Letters The agency posts on its Web site some warning letters that cite companies for making unapproved drugs. None of the 20 largest drugmakers, ranked by U.S. sales, is among those named in the selected enforcement actions on the FDA Web site. One letter was sent to Neil Laboratories Inc. of East Windsor, New Jersey, in May. The FDA accused Neil of selling eight unapproved drugs containing compounds such as guaifenesin, used to treat coughs. Bharat Patel, chief executive officer of Neil, didn't return phone calls seeking comment. The FDA on Oct. 5 said it had negotiated an agreement with Syntho Pharmaceuticals Inc. and Intermax Pharmaceuticals Inc. to stop making unapproved drugs, including cough medicines. The companies, based in Farmingdale, New York, share the same president, Muhammed Malik. He didn't return phone calls seeking comment. Officials at companies that illegally put a drug on the market could face misdemeanor or felony charges, says William Vodra, a former FDA lawyer. The U.S. also could seek to seize drugs or impose other penalties, says Vodra, who now is in private practice in Washington, representing drugmakers. Priorities The regulators say they will focus on unapproved medications that pose safety risks, lack evidence of effectiveness and make deceptive claims. ``We have had to prioritize our enforcement efforts and exercise enforcement discretion,'' in part because resources are limited, the FDA says on its Web site. Some unapproved drugs, such as phenobarbital, used to control seizures, are ``very important therapies'' and appear to help patients, according to the FDA, which says it may not act against beneficial drugs for which alternatives aren't available. A shortage of money and workers has prevented the FDA from doing much about unapproved medicine, says Sasich, the pharmacy professor and co-author of `Worst Pills, Best Pills: A Consumer's Guide to Avoiding Drug-Induced Death or Illness.'' Because of a lack of oversight, companies ``very likely'' could get prescription drugs on the market that prove harmful, he says. `For Their Own Gain' ``You always find people that want to go around regulatory standards for their own gain,'' Sasich says. ``I don't believe that the Food and Drug Administration has the resources to prevent that from happening.'' The FDA had a $1.88 billion budget for the fiscal year ended Sept. 30, and the administration of President George W. Bush has requested $71 million more for fiscal 2007. The FDA needs ``a large boost in funding and staffing,'' the Washington-based Institute of Medicine, which advises the federal government, said in a Sept. 22 report. Physicians, pharmacists and health insurers that pay for prescription medicines say they assume that medications being sold have the government's blessing. Until the FDA decides that drugs need to be removed from the market, the medicines are candidates for insurance reimbursement, says Jeffrey Kelman, chief medical officer for Medicare's Center for Beneficiary Choices. FDA Responsibility FDA officials, not physicians, bear the responsibility for verifying that drugs are approved, says Edward Langston, chairman-elect of the Chicago-based American Medical Association, the nation's largest doctors' group. ``They need to enforce their rules and regulations and not allow these medications to be distributed,'' says Langston, a physician in Lafayette, Indiana. Dozens of drugs not listed in the FDA's database of approved medicines can be found on drugmakers' Web sites and in pharmaceutical directories. Pamlab LLC of Covington, Louisiana, lists several products on its Web site that lack clearance, including Pancof decongestant. ``History has shown that, in general, the products have been safe and effective, or else physicians wouldn't prescribe them,'' says Barry LeBlanc, the company's president and chief operating officer. Some companies' unapproved drugs contain ingredients that were evaluated by the FDA. Unapproved Version That's what happened with carbinoxamine, the antihistamine. The FDA approved a version of the drug in 1953, called Clistin. Fifteen years later, an unapproved allergy drug named Rondec, which contained carbinoxamine and other ingredients, appeared. Cypress Pharmaceutical Inc. of Madison, Mississippi, says it copied Rondec. ``The brand was out there, and had been for many decades at the point where ours came to market,'' also without FDA clearance, says Max Draughn, CEO of Cypress. Makers of unapproved drugs aren't trying to hide from the FDA, says Larry Blansett, CEO of Blansett Pharmacal. The company makes several unapproved medicines, including Cortane- B, which combines an anesthetic, steroid and antibacterial agent to treat ear infections. Blansett and other drugmakers say they have filed required paperwork alerting the FDA of product introductions even for unapproved drugs. Each registered medicine, whether approved or not, carries a 10-digit National Drug Code number, created by the drugmakers and the FDA. The numbers are used by pharmacies to place orders and track inventory. No Secret Ops ``It's not as if these are clandestine operations working under the wire,'' Blansett says. Blansett says he doesn't have to seek approval for drugs with ingredients generally recognized as safe and effective, as he says is the case with Cortane-B, which contains pramoxine, hydrocortisone and chloroxylenol. He nonetheless will do so for that product rather than risk that the government will order it off the market, he says. The approval process may cost the company at least $250,000 and perhaps as much as $20 million, depending on the FDA's demands, the executive says. The FDA may take three to five years to consider the application, he says. In the meantime, he will continue selling the drug. ``Unless the FDA contacts us and tells us it requires anything different, it's OK to market as it is,'' he says. To contact the reporter on this story: Justin Blum in Washington at [email protected] . |



